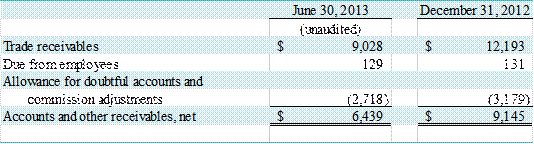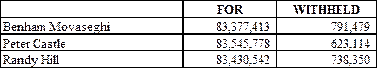NOTE A - ORGANIZATION AND PLAN
OF OPERATIONS
Vasomedical, Inc. was
incorporated in Delaware in July 1987. Unless the context requires otherwise,
all references to “we”, “our”, “us”, “Company”, “registrant”, “Vasomedical” or
“management” refer to Vasomedical, Inc. and its subsidiaries. Until 2010, we were
primarily engaged in designing, manufacturing, marketing and supporting Enhanced
External Counterpulsation (EECP®) systems based on our unique
proprietary technology currently indicated in the United States for use in
cases of stable or unstable angina (i.e., chest pain), congestive heart failure
(“CHF”), acute myocardial infarction (i.e., heart attack, “MI”) and cardiogenic
shock. In May 2010, the Company, through
its wholly-owned subsidiary Vaso Diagnostics, Inc. d/b/a VasoHealthcare, expanded
into the sales representation business via its agreement with GE Healthcare
(“GEHC”), the healthcare business unit of General Electric Company (NYSE: GE), to
be GEHC’s exclusive sales representative for the sale of select GEHC diagnostic
imaging products in specific market segments in the 48 contiguous states of the
United States and the District of Columbia.
In June 2012, the Company entered
into an amendment, effective July 1, 2012, of the sales representative
agreement (“GEHC Agreement”) extending the initial term of three years
commencing July 1, 2010 to five years through June 30, 2015, subject to earlier
termination under certain circumstances.
In September 2011, the Company acquired Fast Growth
Enterprises Limited (FGE), a British Virgin Islands company which owns and
controls two Chinese operating companies - Life Enhancement Technologies Ltd. (“LET”)
and Biox Instruments Co. Ltd. (“Biox”), respectively – to expand its technical and
manufacturing capabilities and to enhance its distribution network, technology,
and product portfolio. Also in September
2011, the Company restructured to further align its business management
structure and long-term growth strategy and now operates through three
wholly-owned subsidiaries. Vaso
Diagnostics d/b/a VasoHealthcare continues as the operating subsidiary for the
sales representation of GE Healthcare diagnostic imaging products; Vasomedical
Global Corp. operates the Company’s Chinese companies; and Vasomedical
Solutions, Inc. manages and coordinates our EECP® therapy business
as well as other medical equipment operations.
We report the operations of Vasomedical Global Corp. and
Vasomedical Solutions, Inc. under our Equipment segment. VasoHealthcare activities are included under
our Sales Representation segment (See Note C).
NOTE B - BASIS OF PRESENTATION AND
CRITICAL ACCOUNTING POLICIES
Basis of Presentation and Use of
Estimates
The accompanying
condensed consolidated financial statements have been prepared in accordance
with accounting principles generally accepted in the United States of America
("U.S. GAAP") and pursuant to the accounting and disclosure rules and
regulations of the Securities and Exchange Commission (the "SEC").
Certain information and disclosures normally included in the condensed
consolidated financial statements prepared in accordance with U.S. GAAP have
been condensed or omitted pursuant to such rules and regulations. Accordingly,
these condensed consolidated financial statements should be read in connection
with the audited consolidated financial statements and related notes thereto
included in the Company's Annual Report on Form 10-K for the year ended December
31, 2012, as filed with the SEC. These condensed consolidated financial
statements include the accounts of the companies over which we exercise
control. In the opinion of management, the accompanying condensed consolidated
financial statements reflect all adjustments (consisting of normal recurring
adjustments) considered necessary for a fair presentation of interim results
for the Company. The results of operations for any interim period are not
necessarily indicative of results to be expected for any other interim period
or the full year.
The
preparation of financial statements in conformity with U.S. GAAP requires
management to make estimates and assumptions that affect the reported amounts
of assets and liabilities as of the date of the condensed
consolidated
financial statements, the disclosure of contingent assets and liabilities in
the condensed consolidated financial statements and the accompanying notes, and
the reported amounts of revenues, expenses and cash flows during the periods
presented. Actual amounts and results could differ from those estimates. The
estimates and assumptions the Company makes are based on historical factors,
current circumstances and the experience and judgment of the Company's
management. The Company evaluates its estimates and assumptions on an ongoing
basis.
Significant Accounting Policies
Note B of the Notes to
Consolidated Financial Statements, included in the Annual Report on Form 10-K
for the year ended December 31, 2012, includes a summary of the significant
accounting policies used in the preparation of the condensed consolidated financial
statements.
Revenue and Expense Recognition for VasoHealthcare
The Company recognizes commission revenue in its Sales
Representation segment (see Note C) when persuasive evidence of an arrangement
exists, service has been rendered, the price is fixed or determinable and
collectability is reasonably assured.
These conditions are deemed to be met when the underlying equipment has
been accepted at the customer site in accordance with the specific terms of the
sales agreement. Consequently, amounts
billable under the agreement with GE Healthcare in advance of the customer
acceptance of the equipment are recorded as accounts receivable and deferred
revenue in the condensed consolidated balance sheets. Similarly, commissions payable to our sales
force related to such billings are recorded as deferred commission expense when
the associated deferred revenue is recorded.
Commission expense is recognized when the corresponding commission
revenue is recognized. Cost of
commissions includes commission expense and, beginning in 2013, costs
associated with the medical device excise tax imposed by the Affordable Care
Act.
Reclassifications
Certain reclassifications have
been made to prior period amounts to conform with the current period presentation.
NOTE C – SEGMENT REPORTING AND
CONCENTRATIONS
The Company views its business in two segments – the Equipment
segment and the Sales Representation segment.
The Equipment segment is engaged in designing, manufacturing, marketing
and supporting EECP® enhanced external counterpulsation systems both
domestically and internationally, as well as the development, production, marketing
and supporting of other medical devices.
The Sales Representation segment operates through the VasoHealthcare
subsidiary and is currently engaged solely in the fulfillment of the Company’s
responsibilities under our agreement with GEHC.
The Company evaluates segment performance based on operating
income. Administrative functions such as
finance, human resources, and information technology are centralized and related
expenses allocated to each segment. Other
costs not directly attributable to operating segments, such as audit, legal,
director fees, investor relations, and others, as well as certain assets –
primarily cash balances – are reported in the Corporate entity below. There are no intersegment revenues. Summary financial information for the
segments is set forth below:
(in thousands) 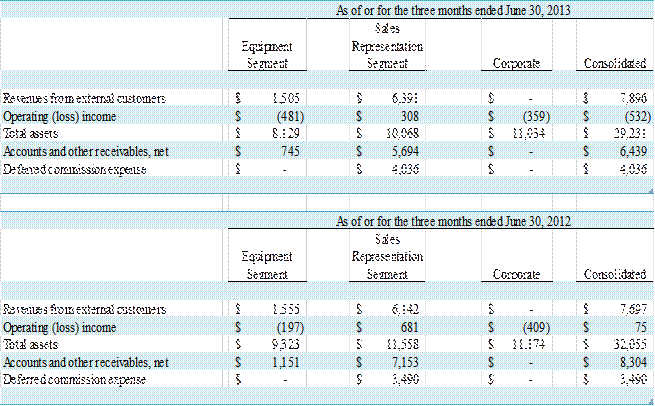
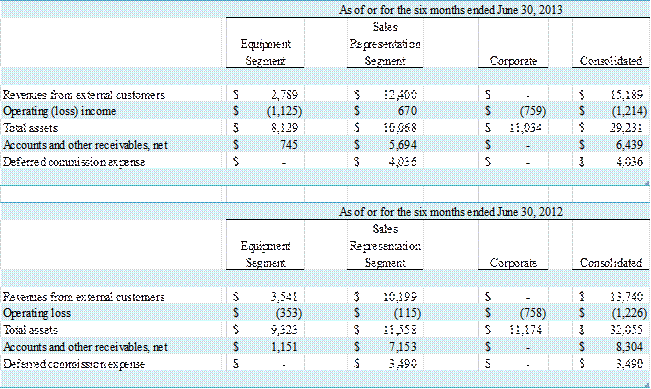
For the three months ended June 30, 2013 and 2012, GE
Healthcare accounted for 81% and 80% of revenue, respectively. For the six months ended June 30, 2013 and 2012,
GE Healthcare accounted for 82% and 74% of revenue, respectively. Also, GE Healthcare accounted for $5.6
million, or 87%, and $8.1 million, or 89%, of accounts and other receivables at
June 30, 2013 and December 31, 2012, respectively.
NOTE D – SHARE-BASED COMPENSATION
The Company complies with ASC
Topic 718 “Compensation – Stock Compensation” (“ASC 718”), which requires all
share-based awards to employees, including grants of employee stock options, to
be recognized in the condensed consolidated financial statements based on their
estimated fair values.
During the three and six month
periods ended June 30, 2013,
the Company granted 340,000 and 400,000 restricted shares of common stock valued
at $63,200 and $74,000, respectively. Shares
valued at $3,600 and $38,000 vested over six month and two year periods,
respectively, with the remainder vesting immediately. During the three month period ended June 30,
2012, the Company granted 2,392,500 shares of restricted common stock valued at
$598,000 in conjunction with the extension of the GEHC Agreement. Such shares vest at various times through
July 1, 2013. In addition, during the
six month period ended June 30, 2012, the Company granted 500,000 shares valued
at $120,000, of which half vested immediately and the remainder vested during
the quarter ended June 30, 2013.
During the three and six month
periods ended June 30, 2013 and 2012,
the Company did not grant any stock options.
Share-based compensation expense
recognized for the three and six month periods ended June 30, 2013 was $133,000
and $271,000, respectively, and $102,000 and $231,000 for the three and six
month periods ended June 30, 2012, respectively. These expenses are included in cost of
revenues; selling, general, and administrative expenses; and research and
development expenses in the condensed consolidated statements of
operations. Expense for share-based consulting
fees with non-employees was $1,000 and $87,000 for the three and six months
ended June 30, 2013, respectively, and $136,000 and $288,000 for the three and
six months ended June 30, 2012, respectively.
Unrecognized expense related to existing share-based compensation and consulting
fees is approximately $176,000 at June 30, 2013 and will be recognized through May
2015.
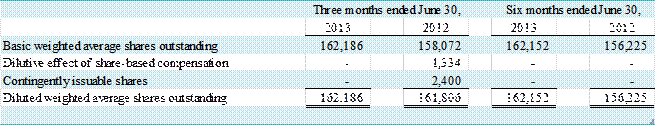
NOTE E – LOSS PER COMMON SHARE
Basic loss per
common share is computed as earnings applicable to common stockholders divided
by the weighted-average number of common shares outstanding for the
period. Diluted loss per common share
reflects the potential dilution that could occur if securities or other
contracts to issue common shares were exercised or converted to common stock.
Diluted earnings per share were computed based on the
weighted average number of shares outstanding plus all potentially dilutive
common stock equivalents. A
reconciliation of basic to diluted shares used in the earnings per share
calculation is as follows:
(in thousands)
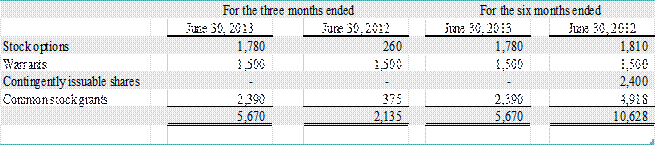
The following table represents
common stock equivalents that were excluded from the computation of diluted
earnings per share for the three and six months ended June 30, 2013 and 2012,
because the effect of their inclusion would be anti-dilutive.
(in thousands)
NOTE
F – FAIR VALUE MEASUREMENTS
The Company complies with the provisions of ASC 820 “Fair
Value Measurements and Disclosures” (“ASC 820”). Under ASC 820, fair value is defined as the
price that would be received to sell an asset or paid to transfer a liability
(i.e., the “exit price”) in an orderly transaction between market participants
at the measurement date.
The following tables present information about the Company’s
assets measured at fair value as of June 30,
2013 and December 31, 2012:
(in thousands)

The fair values of the Company’s cash equivalents invested
in money market funds are determined through market, observable and
corroborated sources.
NOTE
G – ACCOUNTS AND OTHER RECEIVABLES, NET
The following table presents information regarding the
Company’s accounts and other receivables as of June 30, 2013 and December 31,
2012:
(in thousands)
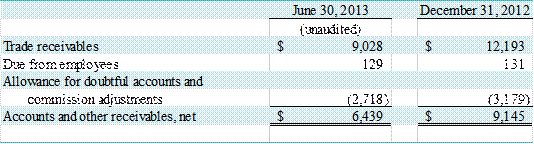
Trade receivables include amounts due for shipped products
and services rendered. Amounts currently
due under the GEHC Agreement are subject to adjustment in subsequent periods
should the underlying sales order amount, upon which the receivable is based,
change.
Allowance for doubtful accounts and commission adjustments
include estimated losses resulting from the inability of our customers to make
required payments, and adjustments arising from subsequent changes in sales
order amounts that may reduce the amount the Company will ultimately receive
under the GEHC Agreement. Due from
employees is primarily commission advances made to sales personnel.
Inventories, net of reserves, consist of the
following: (in
thousands)
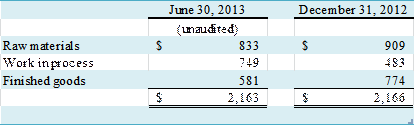
At June 30, 2013 and December
31, 2012, the Company maintained reserves for excess and obsolete inventory of $570,000
and $576,000, respectively.
The changes in the Company’s deferred revenues are as
follows:
(in thousands)

NOTE K – RELATED-PARTY TRANSACTIONS
On June 21, 2007, we entered into a Securities Purchase
Agreement with Kerns Manufacturing Corp. (”Kerns”). Pursuant to this agreement, a five-year
warrant to purchase 4,285,714 shares of our common stock at an initial exercise
price of $0.08 per share was issued to Kerns.
In March 2012, Kerns exercised its warrant and purchased 4,285,714
shares of common stock.
On February 28,
2011, David Lieberman and Edgar Rios were appointed by the Board of
Directors as directors of the Company.
Mr. Lieberman, a practicing attorney in the State of New York, was also
appointed to serve as the Vice Chairman of the Board. He is
currently a senior partner at the law firm of Beckman, Lieberman &
Barandes, LLP, which performs certain legal services for the Company. Fees of approximately
$61,000 and $121,000 were billed by the firm through the three and six months
ended June 30, 2013, respectively, and fees of approximately $67,000 and
$134,000 were billed through the three and six months ended June 30, 2012,
respectively, at which dates no amounts were outstanding.
Mr. Rios
currently is President of Edgary Consultants, LLC, and was appointed a
director in conjunction with the Company’s consulting agreement with Edgary
Consultants, LLC. The consulting agreement
(the “Agreement”) between the Company and Edgary Consultants, LLC
(“Consultant”) commenced on March 1, 2011 and was for a two year term and
expired on February 28, 2013. The Agreement provided for the
engagement of Consultant to assist the Company in seeking broader reimbursement
coverage of EECP® therapy.
In consideration for the services to be
provided by Consultant under the Agreement, the Company agreed to issue to
Consultant or its designees, up to 18,500,000 shares of restricted common
stock of the Company, 3,000,000 of which were issued in March 2011 and the
balance was to be earned on performance. Mr. Lieberman received 600,000 of
these restricted shares. The Company
recorded the fair value of the shares issued to Consultant as a prepaid expense
and amortized the cost ratably over the two year agreement. The unamortized value is reported as deferred
related party consulting expense in our accompanying condensed consolidated balance
sheets as of December 31, 2012. No
performance-based shares were issued and no further compensation is expected to
be paid in conjunction with the agreement.
During the six months ended June 30, 2012, a director
performed consulting services for the Company aggregating approximately $10,000.
Through the Company’s acquisition of FGE in September 2011,
it assumed the liability for $288,000 in unsecured notes payable to the
President of LET and his spouse, of which $95,000 was repaid in December 2011,
and $190,000, bearing interest at 6% per annum, was paid in March 2012. In addition, receivables due from FGE
management aggregating $8,000 and $3,000
were advanced and collected during the three and six months ended June 30, 2013,
respectively, receivables due from FGE management aggregating $4,000 and
$159,000 were collected during the three and six months ended June 30 2012,
respectively.
Biox leases a part of its offices to Genwell Instruments
Co., Ltd., a company partially owned by certain officers of the Company’s
subsidiaries. The lease term is for
twenty months ending December 31, 2014 at a monthly lease amount of
approximately $750.
NOTE L –
STOCKHOLDERS’ EQUITY
Common Stock
See Note K for discussion of common stock issued during the
six months ended June 30, 2012 in connection with related party
agreements. In addition, during the
three and six months ended June 30, 2013 the Company issued 410,313 and 733,054
shares of common stock, respectively, to employees. During the three and six months ended June
30, 2012 the Company issued 500,000 and 562,500 shares of common stock,
respectively, to employees and consultants.
On June 17, 2010 the Board of Directors approved the 2010
Stock Plan (the “2010 Plan”) for officers, directors, employees and consultants
of the Company. The stock issuable under
the 2010 Plan shall be shares of the Company’s authorized but unissued or
reacquired common stock. The maximum
number of shares of common stock which may be issued under the 2010 Plan is
5,000,000 shares.
The 2010 Plan is comprised of two separate equity programs,
the Options Grant Program, under which eligible persons may be granted options
to purchase shares of common stock, and the Stock Issuance Program, under which
eligible persons may be issued shares of common stock directly, either through
the immediate purchase of such shares or as compensation for services rendered
to the Company. The 2010 Plan provides
that the Board of Directors, or a committee of the Board of Directors, will
administer it with full authority to determine the identity of the recipients
of the options or shares and the number of options or shares.
As of June 30, 2013, 3,790,000 restricted shares of common
stock were granted under the 2010 Plan to non-officer employees and consultants
of the Company. As of June 30, 2013, 945,000
shares have been forfeited. In March 2012,
500,000 restricted shares of common stock were granted under the 2010 Plan to
an officer, of which 250,000 vested immediately with the remainder vesting over
a one year period. In June 2012, 2,392,500
additional restricted common shares, vesting at various times through July 1,
2013, were granted to non-officer employees in conjunction with the extension
of the GEHC Agreement, of which 373,500 shares had been forfeited as of June 30,
2013.
No options were issued under the 2010 Plan during the three
and six month periods ended June 30, 2013 and 2012.
In April 2013, the Company’s Board of Directors authorized
a share repurchase program of up to $1.5 million, which was subsequently
increased in July 2013 to $2.0 million, of the Company’s common stock. As of June 30, 2013, 1,005,998 shares had
been repurchased at a cost of $182,000, which has been recorded as treasury
stock in the accompanying condensed consolidated balance sheet as of June 30,
2013. As of August 9, 2013 a total of 7,989,258
shares at a cost of $1,347,197 have been repurchased. (See Note O – Subsequent Events.)
Preferred Stock
Pursuant to its conversion terms, all Series E preferred
stock was deemed automatically converted to common stock effective July 1, 2011,
and all converted shares were issued as of June 30, 2012.
NOTE M – COMMITMENTS
AND CONTINGENCIES
Sales representation agreement
In June 2012, the Company concluded an
amendment of the GEHC Agreement with GE Healthcare, originally signed on May
19, 2010. The amendment, effective July
1, 2012, extended the initial term of three years commencing July 1, 2010 to
five years through June 30, 2015, subject to earlier termination under certain
circumstances. These circumstances
include not materially achieving certain sales goals, not maintaining a minimum
number of sales representatives, and various legal and GEHC policy
requirements. Under the terms of the
agreement, the Company is required to lease dedicated computer equipment from GEHC
for connectivity to their network.
In conjunction with the extension of the GEHC
Agreement, the Company granted VasoHealthcare employees both stock and
cash-based performance incentives for the ensuing year. The incentives provide for cash payments of
up to $2.4 million and 2.4 million shares of restricted common stock grants and
vest at various times through July 1, 2013.
A condition of the incentives is that the employees remain continuously
employed through the vesting dates. As
of June 30, 2013, approximately $0.5 million and 0.5 million shares remained
unvested. These amounts vested on July
1, 2013.
NOTE N - RECENTLY ISSUED ACCOUNTING
PRONOUNCEMENTS
Adoption of New Standards
Other Comprehensive Income:
Presentation of Comprehensive Income
In February 2013, new guidance was issued that amends the
current comprehensive income guidance.
The new guidance requires entities to disclose the effect of each item
that was reclassified in its entirety out of accumulated other comprehensive
income and into net income on each affected net income line item. For reclassification items that are not
reclassified in their entirety into net income, a cross-reference to other
required disclosures is required. The new guidance is to be applied
prospectively for annual reporting periods beginning after December 15, 2012
and interim periods within those years.
The adoption of this new guidance did not have an impact on the Company’s
consolidated financial position, results of operations, or cash flows.
NOTE O –
SUBSEQUENT EVENT
As discussed in Note L, on
July 30, 2013 the Company approved an increase to its previously announced
stock repurchase program of its common stock, from up to $1.5 million to up to
$2 million. As of the close of business
on August 9, 2013, the Company has acquired 7,989,258 shares of its common
stock pursuant to this program, including a block transaction of 6,708,134
shares on July 30, 2013, at an aggregate cost to the Company of $1,347,197.
Except for
historical information contained in this report, the matters discussed are
forward-looking statements that involve risks and uncertainties. When used in
this report, words such as “anticipates”, “believes”, “could”, “estimates”,
“expects”, “may”, “plans”, “potential” and “intends” and similar expressions,
as they relate to the Company or its management, identify forward-looking
statements. Such forward-looking statements are based on the beliefs of the
Company’s management, as well as assumptions made by and information currently
available to the Company’s management. Among the factors that could cause
actual results to differ materially are the following: the effect of business and economic conditions; the effect of the dramatic changes taking place in the
healthcare environment; the impact of competitive procedures and products and
their pricing; medical insurance reimbursement policies; unexpected
manufacturing or supplier problems; unforeseen difficulties and delays in the
conduct of clinical trials and other product development programs; the actions
of regulatory authorities and third-party payers in the United States and
overseas; uncertainties about the acceptance of a novel therapeutic modality by
the medical community; continuation of the GEHC Agreement and the risk factors
reported from time to time in the Company’s SEC reports, including its recent
report on Form 10-K. The Company
undertakes no obligation to update forward-looking statements as a result of
future events or developments.
General Overview
Vasomedical, Inc. was incorporated in Delaware in July
1987. Unless the context requires
otherwise, all references to “we”, “our”, “us”, “Company”, “registrant”,
“Vasomedical” or “management” refer to Vasomedical, Inc. and its subsidiaries. Until 2010, we were primarily engaged in
designing, manufacturing, marketing and supporting Enhanced External
Counterpulsation (EECP®) systems, based on our proprietary
technology, to physicians and hospitals throughout the United States and in select
international markets.
In May 2010, the Company, through its wholly-owned
subsidiary Vaso Diagnostics, Inc. d/b/a VasoHealthcare, expanded into the sales
representation business via its agreement with GE Healthcare (“GEHC”), the
healthcare business unit of General Electric Company (NYSE: GE), to be GEHC’s
exclusive sales representative for the sale of select GEHC diagnostic imaging
products in specific market segments in the 48 contiguous states of the United
States and the District of Columbia. In June 2012, the Company
entered into an amendment, effective July 1, 2012, of the sales representative
agreement (“GEHC Agreement”) extending the initial term of three years
commencing July 1, 2010 to five years through June 30, 2015, subject to earlier
termination under certain circumstances.
In September 2011, the Company acquired Fast Growth
Enterprises Limited (FGE), a British Virgin Islands company, which owns and
controls two Chinese operating companies - Life Enhancement Technology Ltd. and
Biox Instruments Co. Ltd., respectively - to expand its technical and
manufacturing capabilities and to enhance its distribution network, technology,
and product portfolio. Also in September
2011, the Company restructured to further align its business management structure
and long-term growth strategy, and now operates through three wholly-owned
subsidiaries. Vaso Diagnostics d/b/a
VasoHealthcare continues as the operating subsidiary for the sales
representation of GE diagnostic imaging products; Vasomedical Global Corp.
operates the Company’s Chinese companies; and Vasomedical Solutions, Inc. was
formed to manage and coordinate our EECP® therapy business as well
as other medical equipment operations.
We now report the operations of Vasomedical Global Corp.
and Vasomedical Solutions, Inc. under our Equipment segment. VasoHealthcare activities are included under
our Sales Representation segment (see Note C).
The Company expects to achieve profitability
through reaching a higher commission rate under the GEHC Agreement, growth in
our China operations and by expanding our product portfolio. In addition, the Company plans to pursue
other accretive acquisitions in the international and domestic markets and to
expand our sales representation business.
Critical Accounting Policies and Estimates
Our discussion and analysis of
our financial condition and results of operations are based upon the
accompanying unaudited condensed consolidated financial statements, which have
been prepared in accordance with accounting principles generally accepted in
the United States (“U.S. GAAP”). The preparation of financial statements in
conformity with U.S. GAAP requires management to make judgments, estimates and
assumptions that affect the reported amounts of assets, liabilities, revenue,
expenses, and the related disclosures at the date of the financial statements
and during the reporting period. Although these estimates are based on our
knowledge of current events, our actual amounts and results could differ from
those estimates. The estimates made are based on historical factors, current
circumstances, and the experience and judgment of our management, who
continually evaluate the judgments, estimates and assumptions and may employ
outside experts to assist in the evaluations.
Certain of our accounting
policies are deemed “critical”, as they are both most important to the
financial statement presentation and require management’s most difficult,
subjective or complex judgments as a result of the need to make estimates about
the effect of matters that are inherently uncertain. For a discussion of our
critical accounting policies, see “Management’s Discussion and Analysis of
Financial Condition and Results of Operations” in our Annual Report on Form 10-K
for the year ended December 31, 2012.
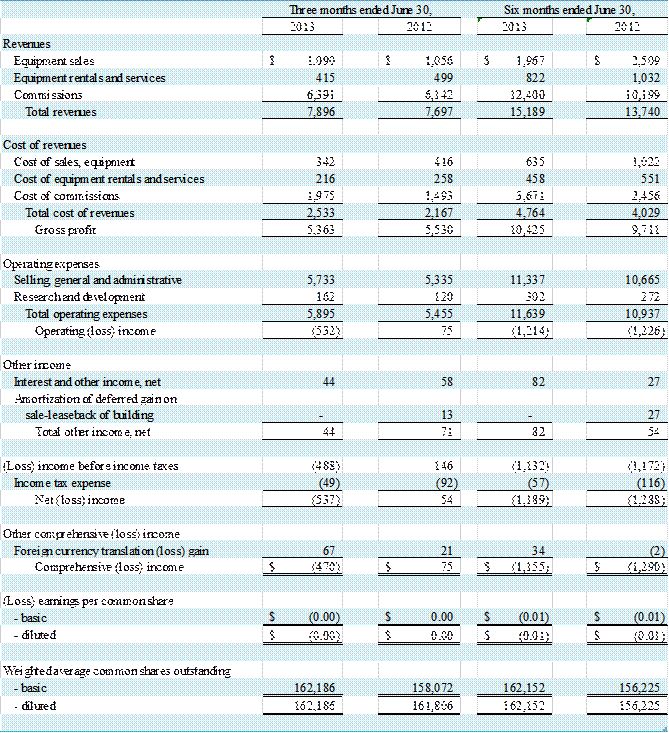
Results of Operations – For the Three Months Ended June 30,
2013 and 2012
Total revenue for the three months ended June 30, 2013 and
2012 was $7,896,000 and $7,697,000, respectively, an increase of $199,000, or 3%.
Net loss for the three months ended June 30, 2013 was $537,000 compared to net income
of $54,000 for the three months ended June 30, 2012. The change from net income to net loss was
primarily attributable to a $167,000 decrease in gross profit and a $398,000 increase
in selling, general and administrative costs.
Our total net (loss)/earnings was $(0.00) and $0.00 per basic and
diluted common share for the three months ended June 30, 2013 and 2012,
respectively.
Revenues
Revenue in our Equipment segment
decreased by $50,000, or 3%, to $1,505,000 for the three-month period ended June
30, 2013 from $1,555,000 for the same period of the prior year. Equipment segment revenue from equipment
sales increased by $34,000, or 3%, to $1,090,000 for the three-month period
ended June 30, 2013 as compared
to $1,056,000 for the same period in the prior year primarily due to a $310,000
increase in international sales by our China operations, partially offset by a
decrease of $283,000 in EECP® revenues as a result of lower sales volume.
Current demand for EECP®
systems domestically will likely remain soft until there is greater clinical
acceptance for the use of EECP® therapy in treating patients with
angina or angina equivalent symptoms who meet the current reimbursement
guidelines, or a favorable change in current reimbursement policies by CMS or
third party payers to consider EECP therapy as a first-line treatment option
for angina or cover some or all Class II & III heart failure patients.
Patients with angina or angina equivalent symptoms eligible for reimbursement
under current policies include many with serious comorbidities, such as heart
failure, diabetes, peripheral vascular disease and/or others.
Equipment segment revenue from
equipment rental and services decreased 17% to $415,000 in the second quarter
of 2013 from $499,000 in the second quarter of 2012. Revenue from equipment
rental and services represented 28% and 32% of total Equipment segment revenue
in the second quarters of fiscal 2013 and fiscal 2012, respectively. The decrease in revenue generated from
equipment rentals and services is due primarily to decreased accessory part and
service contract revenues.
Commission revenues in the Sales
Representation segment were $6,391,000 in the second quarter of 2013, as
compared to $6,142,000 in the second quarter of 2012, an increase of 4%. The increase in commission revenue in the second
quarter of 2013 is due primarily to an increase in volume of equipment
delivered by GEHC. The Company
recognizes revenue when the underlying equipment has been accepted at the
customer site in accordance with the specific terms of the sales
agreement. Consequently, amounts
billable under the agreement with GE Healthcare prior to customer acceptance of
the equipment are recorded as deferred revenue in the condensed consolidated balance
sheet. As of June 30, 2013, $12,076,000
in deferred commission revenue was recorded in the Company’s condensed
consolidated balance sheet, of which $2,542,000 is long-term. At June 30, 2012, $13,896,000 in deferred commission
revenue was recorded in the Company’s condensed consolidated balance sheet, of
which $4,748,000 was long-term.
Gross Profit
The Company had a gross profit
of $5,363,000 in the second quarter of 2013 compared to $5,530,000 in the second
quarter of the prior year, a decrease of 3%.
The decrease is principally due to lower commission rates on equipment
orders in 2012 which were delivered in the second quarter of 2013 in the Sales
Representation segment. Equipment
segment gross profit increased to $947,000, or 63% of Equipment segment
revenues, for the second quarter of 2013 compared to $881,000, or 57% of
Equipment segment revenues, for the same quarter of 2012. Equipment segment gross profit improved due to
a greater proportion of other medical equipment sales versus EECP®
equipment sales, which have higher margins than EECP® equipment, in
the second quarter of 2013. Gross profit
margin on EECP® equipment improved as a result of lower production costs
arising from the acquisition of LET, our primary supplier of certain EECP®
systems. Gross profit in the Equipment
segment is dependent on a number of factors, particularly the mix of new and
refurbished EECP® systems and the mix of models sold, their
respective average selling prices, the mix of EECP® units sold,
rented or placed during the period, the ongoing costs of servicing EECP®
systems, and certain fixed period costs, including facilities, payroll and
insurance.
Sales Representation segment
gross profit was $4,416,000, or 69%, for the three months ended June 30, 2013
as compared to $4,649,000, or 76%, for the three months ended June 30, 2012. The decrease was due to lower commission
rates, coupled with commission expense which is at the same rate as in 2012, partially
offset by higher revenue in this segment.
Cost of commissions of $1,975,000 and $1,493,000, for the three months
ended June 30, 2013 and 2012, respectively, reflects commission expense
associated with recognized commission revenues, and, starting in 2013,
additional costs associated with the medical device excise tax imposed by the Affordable
Care Act. Commission expense associated
with deferred revenue is recorded as deferred commission expense until the
related commission revenue is earned.
Operating (Loss) Income
Operating loss was $532,000 for the three months ended June
30, 2013 as compared to operating income of $75,000 for the three months ended June
30, 2012, a decrease of $607,000. The decrease
in operating income was primarily attributable to the decrease in gross profit
in the Sales Representation segment as described above, where operating income
was $308,000 for the second quarter of 2013 compared to operating income of $681,000
in the same quarter of the prior year. Equipment
segment operating loss also increased by $284,000 to $481,000 in the second
quarter of 2013 from $197,000 in the same period of the prior year.
Selling, general and
administrative (“SG&A”) expenses for the second quarter of 2013 and 2012 were
$5,733,000, or 73% of revenues, and $5,335,000, or 69% of revenues,
respectively, reflecting an increase of $398,000 or approximately 7%. The
increase in SG&A expenditures in the second quarter of 2013 resulted
primarily from increased costs associated with the expansion of the Equipment
segment sales team of approximately $200,000, as well as $140,000 higher costs in
the Sales Representation segment, which includes $76,000 of non-recurring costs
attributable to the renewal of the GEHC contract.
Research
and development (“R&D”) expenses of $162,000, or 2% of revenues (11% of
Equipment segment revenues), for the second quarter of 2013 increased by $42,000,
or 35%, from $120,000, or 2% of revenues (8% of Equipment segment revenues),
for the second quarter of 2012. The increase is primarily attributable to an increase
in product development expenses.
Interest and Other Income, Net
Interest and other income
(expense), net for the second quarter of 2013 was $44,000 as compared to $58,000
for the second quarter of 2012. The decrease was due primarily to lower
interest earnings on the Company’s cash balances.
Amortization of Deferred Gain on Sale-leaseback of Building
The amortization of the deferred gain on the sale-leaseback of our Westbury, NY facility for
the second quarter of 2012 was $13,000. The
deferred gain was fully amortized in the third quarter of 2012.
Income Tax Expense
During
the second quarter of 2013 we recorded an income tax expense of $49,000 as
compared to an income tax expense of $92,000 for the second quarter of 2012. The decrease arose from reduced tax expense
at our Chinese subsidiaries.
Results of Operations – For the Six Months Ended June 30,
2013 and 2012
Total revenue for the six months ended June 30, 2013 and
2012 was $15,189,000 and $13,740,000, respectively, an increase of $1,449,000,
or 11%. Net loss for the six months ended June 30, 2013 was $1,189,000 compared
to a net loss of $1,288,000 for the six months ended June 30, 2012. The decrease in net loss was primarily
attributable to a $714,000 increase in gross profit, partially offset by a $672,000
increase in selling, general and administrative costs. Our total net loss was $0.01 per basic and
diluted common share for the six months ended June 30, 2013 and 2012,
respectively.
Revenues
Revenue in our Equipment segment
decreased by $752,000, or 21%, to $2,789,000 for the six-month period ended June
30, 2013 from $3,541,000 for the same period of the prior year. Equipment segment revenue from equipment
sales decreased by $542,000, or 22%, to $1,967,000 for the six-month period
ended June 30, 2013 as compared
to $2,509,000 for the same period in the prior year primarily due to a decrease
of $806,000 in EECP® revenues as a result of lower sales volume.
Current demand for EECP®
systems domestically will likely remain soft until there is greater clinical
acceptance for the use of EECP® therapy in treating patients with
angina or angina equivalent symptoms who meet the current reimbursement
guidelines, or a favorable change in current reimbursement policies by CMS or
third party payers to consider EECP therapy as a first-line treatment option
for angina or cover some or all Class II & III heart failure patients.
Patients with angina or angina equivalent symptoms eligible for reimbursement
under current policies include many with serious comorbidities, such as heart
failure, diabetes, peripheral vascular disease and/or others.
Equipment segment revenue from
equipment rental and services decreased 20% to $822,000 in the first half of
2013 from $1,032,000 in the first half of 2012. Revenue from equipment rental
and services represented 29% of total Equipment segment revenue in the first
six months of both fiscal 2013 and fiscal 2012.
The decrease in revenue generated from equipment rentals and services is
due primarily to decreased accessory part and service contract revenues.
Commission revenues in the Sales
Representation segment were $12,400,000 in the first half of 2013, as compared
to $10,199,000 in the first half of 2012, an increase of 22%. The increase in commission revenue in the
first half of 2013 is due primarily to an increase in volume of equipment
delivered by GEHC. The Company
recognizes revenue when the underlying equipment has been accepted at the
customer site in accordance with the specific terms of the sales
agreement. Consequently, amounts
billable under the agreement with GE Healthcare prior to customer acceptance of
the equipment are recorded as deferred revenue in the condensed consolidated
balance sheet. As of June 30, 2013, $12,076,000
in deferred commission revenue was recorded in the Company’s condensed
consolidated balance sheet, of which $2,542,000 is long-term. At June 30, 2012, $13,896,000 in deferred
commission revenue was recorded in the Company’s condensed consolidated balance
sheet, of which $4,748,000 was long-term.
Gross Profit
The Company had a gross profit
of $10,425,000 in the first half of 2013 compared to $9,711,000 in the first half
of the prior year, an increase of 7%. The
increase is principally due to the increase in commission revenue discussed
above. Equipment segment gross profit decreased
to $1,696,000, or 61% of Equipment segment revenues, for the first half of 2013
compared to $1,968,000, or 56% of Equipment segment revenues, for the same period
of 2012. Equipment segment gross profit declined
due to lower equipment sales in the first half of 2013. Gross profit margin on EECP®
equipment improved as a result of lower production costs arising from the
acquisition of LET, our primary supplier of certain EECP® systems. Gross profit in the Equipment segment is
dependent on a number of factors, particularly the mix of new and refurbished
EECP® systems and the mix of models sold, their respective average
selling prices, the mix of EECP® units sold, rented or placed during
the period, the ongoing costs of servicing EECP® systems, and
certain fixed period costs, including facilities, payroll and insurance.
Sales Representation segment
gross profit was $8,729,000, or 70%, for the six months ended June 30, 2013 as
compared to $7,743,000, or 76%, for the six months ended June 30, 2012. The increase was due to higher revenue in
this segment, as explained above, partially offset by lower commission rates in
the first half of 2013. Cost of
commissions of $3,671,000 and $2,456,000, for the six months ended June 30,
2013 and 2012, respectively, reflects commission expense associated with
recognized commission revenues, and, starting in 2013, additional costs
associated with the medical device excise tax imposed by the Affordable Care
Act. Commission expense associated with
deferred revenue is recorded as deferred commission expense until the related
commission revenue is earned.
Operating (Loss) Income
Operating loss was $1,214,000 for the six months ended June
30, 2013 as compared to an operating loss of $1,226,000 for the six months
ended June 30, 2012, an improvement of $12,000.
The decrease in operating loss was primarily attributable to improved
operating performance in the Sales Representation segment, where operating income
was $670,000 for the first half of 2013 compared to an operating loss of $115,000
in the same period of the prior year. Partially
offsetting the improvement in the Sales Representation segment was an increase
in operating loss of $772,000 in the Equipment segment to $1,125,000 in the
first half of 2013 from an operating loss of $353,000 in the same period of the
prior year.
Selling, general and
administrative (“SG&A”) expenses for the first half of 2013 and 2012 were $11,337,000,
or 75% of revenues, and $10,665,000, or 78% of revenues, respectively,
reflecting an increase of $672,000 or approximately 6%. The increase in
SG&A expenditures in the first half of 2013 resulted primarily from
increased costs associated with the expansion of the Equipment segment sales
team of approximately $320,000, as well as $201,000 higher costs in the Sales
Representation segment. The increase of
$201,000 includes $314,000 of non-recurring costs attributable to the renewal
of the GEHC contract, partially offset by reductions in other areas.
Research
and development (“R&D”) expenses of $302,000, or 2% of revenues (11% of
Equipment segment revenues), for the first half of 2013 increased by $30,000,
or 11%, from $272,000, or 2% of revenues (8% of Equipment segment revenues),
for the first half of 2012. The increase is primarily attributable to an increase
in product development expenses.
Interest and Other Income, Net
Interest and other income,
net for the first half of 2013 was $82,000 as compared to $27,000 for the first
half of 2012. The improvement was due to certain non-recurring non-operating
expenses incurred by our Chinese subsidiaries in the first half of 2012.
Amortization of Deferred Gain on Sale-leaseback of Building
The amortization of the deferred gain on the sale-leaseback of our Westbury, NY facility for
the first half of 2012 was $27,000. The
deferred gain was fully amortized in the third quarter of 2012.
Income Tax Expense
During the first half of 2013 we
recorded income tax expense of $57,000 as compared to income tax expense of $116,000
for the first half of 2012. The decrease
resulted from lower tax expense at our Chinese subsidiaries.
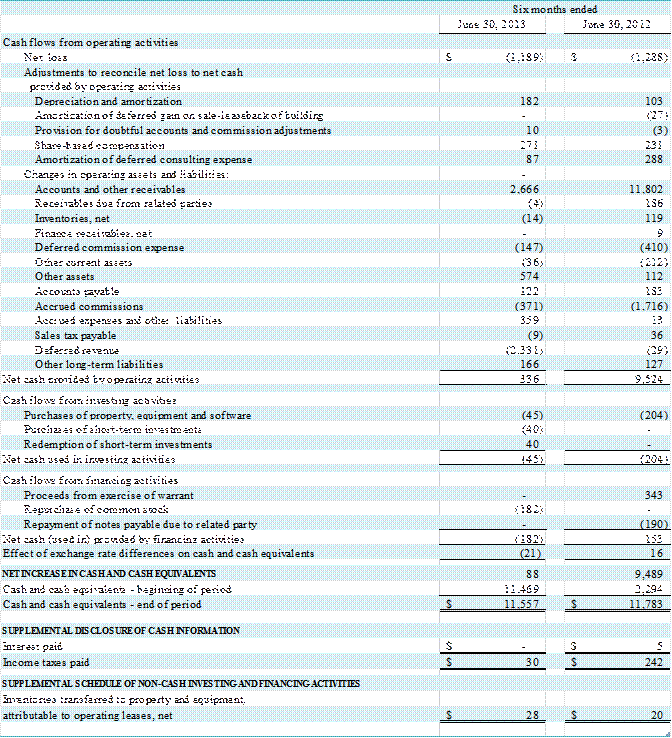
Liquidity and Capital Resources
Cash and Cash Flow
We have financed our operations
from working capital. At June 30, 2013,
we had cash and cash equivalents of $11,557,000, short-term investments of $111,000
and working capital of $5,195,000 compared to cash and cash equivalents of $11,469,000,
short-term investments of $110,000 and working capital of $7,538,000 at
December 31, 2012.
Cash provided by operating
activities was $336,000 during the first six months of 2013, which consisted of
a net loss after adjustments to reconcile net loss to net cash of $639,000,
offset by cash provided by operating assets and liabilities of $975,000. The
changes in the account balances primarily reflect a decrease in accounts and
other receivables of $2,666,000, partially offset by decreases in deferred
revenue of $2,331,000 and accrued commissions of $371,000. Under the GEHC Agreement the Company earns
progressively higher commission rates as calendar year targets are met, and
this commission structure has a significant impact on our cash flows. As we
achieve these targets, the higher commission rates are retroactively applied to
all sales in the calendar year, and therefore, the significantly higher
commission billings and recognized revenue generated in the fourth quarter of
2011 resulted in significant cash inflows in the first half of 2012. As we previously reported in our Annual
Report on Form 10-K for the year ended December 31, 2012, lower commission
rates were earned in 2012 than in 2011, resulting in lower cash inflows in the
first half of 2013.
Cash used in investing activities during the six-month period
ended June 30, 2013 was $45,000 for the
purchase of equipment and software.
Cash used in financing activities during the six-month
period ended June 30, 2013 was $182,000 for the repurchase of common stock.
Liquidity
The Company expects to achieve
profitability and continued positive cash flow through reaching a higher
commission rate under the GEHC Agreement, growth in our China operations and by
expanding our product portfolio. In
addition, the Company plans to pursue other accretive acquisitions in the
international and domestic markets and to expand our sales representation
business.
While we expect to generate
positive operating cash flows for 2013, the progressive nature of the
commission rates under the GEHC Agreement can cause related cash inflows to
vary widely during the year.
Evaluation of Disclosure Controls and Procedures
Disclosure
controls and procedures reporting as promulgated under the Exchange Act is
defined as controls and procedures that are designed to ensure that information
required to be disclosed by us in the reports that we file or submit under the
Exchange Act are recorded, processed, summarized and reported within the time
periods specified in the SEC rules and forms.
Disclosure controls and procedures include without limitation, controls
and procedures designed to ensure that information required to be disclosed by
us in the reports that we file or submit under the Exchange Act is accumulated
and communicated to our management, including our Chief Executive Officer
(“CEO”) and Chief Financial Officer (“CFO”), or persons performing similar
functions, as appropriate to allow timely decisions regarding required
disclosure.
Our CEO and our CFO have
evaluated the effectiveness of the design and operation of our disclosure
controls and procedures as of June 30, 2013 and have concluded that the
Company’s disclosure controls and procedures were not effective as of June 30,
2013 due to insufficient controls and management review over the recording of
certain transactions, and the lack of accounting personnel with appropriate
level of knowledge and experience in accounting principles generally accepted
in the United States of America and related accounting systems and the closing
process at our China subsidiaries. The
Company has engaged additional accounting personnel, and has implemented a
review process of its closing procedures, related disclosures, and the approval
of certain transactions, and expect that these actions have resolved the
issues. Management expects to test the
effectiveness of these modifications in connection with the 2013 audit..
Changes in Internal Control Over Financial Reporting
There
was no change in the Company’s internal control over financial reporting during
the Company’s last fiscal quarter that has materially affected, or is
reasonably likely to materially affect, the Company’s internal control over
financial reporting.
On July 10, 2013, Vasomedical, Inc.
(the “Company”) held its 2013 Annual Meeting of Shareholders (the “Annual
Meeting”). At the Annual Meeting, shareholders of record on May 17,
2013 were entitled to vote 163,247,623 shares of the Company’s common stock
(the “Common Stock”). A total of 140,576,777 shares of Common Stock
(86.4% of all shares entitled to vote at the Annual Meeting) were represented
at the Annual Meeting in person or by proxy.
At the Annual Meeting, the
shareholders of the Company (i) elected three Class II director nominees to
hold office until the 2016 Annual Meeting of Shareholders and until their
successors are elected, (ii) ratified the appointment of Rothstein Kass &
Company, P.C. as the Company’s independent registered public accountants for
the fiscal year ended December 31, 2013, (iii) approved an advisory,
non-binding resolution regarding the compensation of the Company’s named
executive officers, and (iv) approved every three years in an advisory,
non-binding resolution as the frequency of the advisory vote on the
compensation of the Company’s named executive officers. Set forth
below are the final voting results for each of the proposals submitted to a
vote of the shareholders.
Election of Directors
The shareholders of the Company
elected each of the director nominees proposed by the Company’s Board of
Directors. The voting results were as follows:
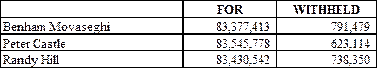
Ratification of Appointment of Independent Registered
Public Accountants
The shareholders of the Company
ratified the appointment of Rothstein Kass & Company, P.C. as the Company’s
independent registered public accountants for the fiscal year ended December
31, 2013. The voting results were as follows:
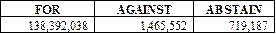
Advisory Vote on Executive Compensation
The shareholders of the Company
approved, on an advisory basis, the resolution regarding the compensation of
the Company’s named executive officers. The voting results were as
follows:
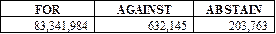
Advisory Vote on the Frequency of Votes on Executive
Compensation
The shareholders of the Company
approved, on an advisory basis, the holding of advisory votes on executive
compensation every three years. The voting results were as follows:
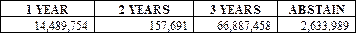
Exhibits
31 Certifications of the Chief Executive
Officer and the Chief Financial Officer pursuant to Rules 13a-14(a) as adopted
pursuant to Section 302 of the Sarbanes-Oxley Act of 2002.
32 Certifications of the Chief Executive
Officer and the Chief Financial Officer pursuant to 18 U.S.C. Section 1350 as
adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002.
In accordance with the requirements of the Exchange Act, the
Registrant caused this report to be signed on its behalf by the undersigned,
thereunto duly authorized.
VASOMEDICAL,
INC.
By: /s/ Jun Ma
Jun Ma
President
and Chief Executive Officer
(Principal
Executive Officer)
/s/ Michael J. Beecher .
Michael
J. Beecher
Chief
Financial Officer and Principal Accounting Officer
Date: August 13, 2013
![]()